⏱️ 7 min read
Quantum physics represents one of the most fascinating and mind-bending fields of science, challenging our everyday understanding of reality. This branch of physics explores the behavior of matter and energy at the smallest scales—atoms and subatomic particles—where the rules that govern our macroscopic world no longer apply. From particles existing in multiple places simultaneously to instantaneous connections across vast distances, the quantum realm is filled with phenomena that seem more like science fiction than science fact. Here are ten remarkable facts that showcase just how strange and wonderful quantum physics truly is.
Exploring the Bizarre World of Quantum Mechanics
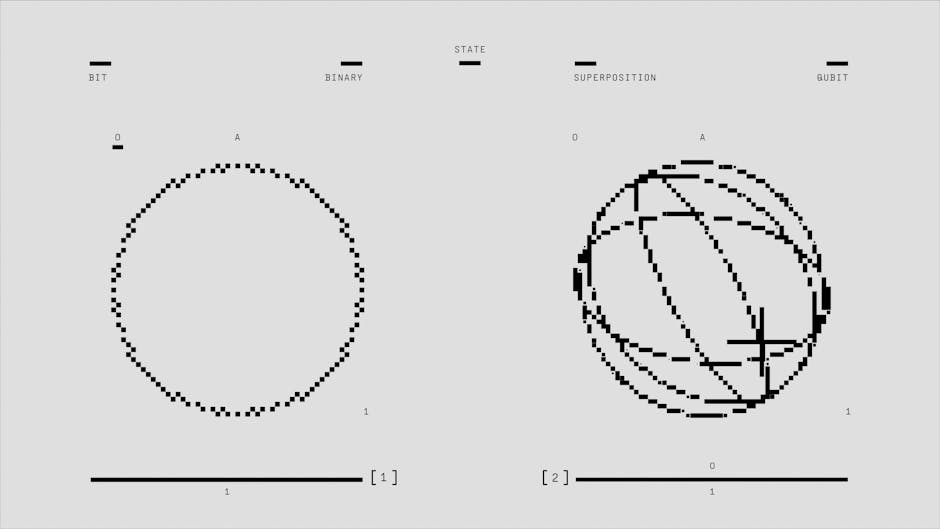
1. Particles Can Exist in Multiple States Simultaneously
One of the most counterintuitive principles in quantum physics is superposition, which states that particles can exist in multiple states at the same time until they are observed or measured. The famous thought experiment known as Schrödinger's cat illustrates this concept: a cat in a sealed box with a quantum-triggered poison can theoretically be both alive and dead simultaneously until someone opens the box to check. In the quantum world, electrons can spin in multiple directions, be in different energy levels, or even exist in several locations at once. This phenomenon isn't just theoretical—it's been repeatedly demonstrated in laboratory experiments and forms the foundation of emerging quantum computing technology.
2. Observation Fundamentally Changes Reality
The act of observing or measuring a quantum system fundamentally alters its state, a phenomenon known as the observer effect. This isn't about the physical disturbance caused by measurement tools; rather, it's a fundamental feature of quantum mechanics. Before measurement, particles exist in a probabilistic state described by a wave function. The moment we observe them, this wave function "collapses" into a definite state. This has profound philosophical implications, suggesting that at the quantum level, reality itself is influenced by observation. The double-slit experiment dramatically demonstrates this effect, showing how electrons behave differently when watched versus when unobserved.
3. Quantum Entanglement Creates Instant Connections Across Space
Albert Einstein famously called it "spooky action at a distance," but quantum entanglement is a well-established phenomenon where two particles become correlated in such a way that measuring one instantly affects the other, regardless of the distance separating them. When particles become entangled, they form a single quantum system where the state of one particle is intrinsically linked to the state of its partner. This connection persists even if the particles are separated by billions of miles. Scientists have experimentally verified entanglement over distances exceeding 1,200 kilometers, and this phenomenon is now being harnessed for quantum communication and cryptography applications.
4. Electrons Can Tunnel Through Impenetrable Barriers
In the quantum world, particles can pass through barriers that would be impossible to cross according to classical physics—a phenomenon called quantum tunneling. This occurs because particles at the quantum level are also described by waves, and these waves can extend beyond physical barriers. While the probability of tunneling decreases with barrier thickness, it never reaches absolute zero. This seemingly impossible phenomenon is actually essential to many everyday technologies. Nuclear fusion in stars, including our sun, relies on quantum tunneling to overcome the electromagnetic repulsion between atomic nuclei. Scanning tunneling microscopes use this effect to image individual atoms, and modern electronic devices like flash memory depend on it for operation.
5. The Heisenberg Uncertainty Principle Sets Fundamental Limits on Knowledge
Werner Heisenberg discovered that there are fundamental limits to how precisely we can simultaneously know certain pairs of properties of a particle, such as its position and momentum. This isn't a limitation of our measurement tools but a fundamental feature of nature itself. The more precisely we determine a particle's position, the less precisely we can know its momentum, and vice verса. This uncertainty isn't negligible—it's built into the fabric of reality at the quantum scale. This principle has profound implications, suggesting that at the most fundamental level, the universe is inherently probabilistic rather than deterministic, contradicting the classical physics view that everything could theoretically be predicted with sufficient information.
6. Quantum Particles Have No Definite Properties Until Measured
Unlike everyday objects that have definite properties whether we're looking at them or not, quantum particles don't possess definite characteristics until they're measured. Before measurement, they exist in a state of probability—they have a certain likelihood of being found with various properties, but no actual defined values. A photon doesn't have a definite polarization, and an electron doesn't have a definite spin direction until these properties are measured. This isn't merely that we don't know these properties; they genuinely don't exist in a definite state. This fundamental indeterminacy is what makes quantum mechanics so philosophically challenging and fundamentally different from our everyday experience of reality.
7. Quantum Physics Enables Particles to Be in Two Places at Once
The wave-particle duality of quantum mechanics means that particles can literally be in multiple locations simultaneously. Experiments have shown that individual atoms, and even molecules containing thousands of atoms, can traverse two different paths at the same time. In one remarkable experiment, scientists demonstrated that a molecule called buckminsterfullerene (composed of 60 carbon atoms) could pass through two separate slits simultaneously, creating an interference pattern that proved it traveled both paths. This isn't about not knowing which path the particle took—the particle genuinely takes both paths. This property is being exploited in quantum computing, where quantum bits can process multiple calculations simultaneously by existing in multiple states at once.
8. The Quantum Vacuum Isn't Actually Empty
What we think of as empty space—a perfect vacuum—is actually teeming with activity at the quantum level. The Heisenberg uncertainty principle means that even in a vacuum, particle-antiparticle pairs are constantly popping into existence and annihilating each other in incredibly brief moments. These "virtual particles" aren't just theoretical constructs; they have measurable effects on real particles and are responsible for phenomena like the Casimir effect, where two metal plates placed very close together in a vacuum experience an attractive force due to virtual particles. This quantum vacuum energy permeates all of space and may be related to the mysterious dark energy that's accelerating the universe's expansion.
9. Quantum Mechanics Explains Why Matter Doesn't Collapse
Without quantum mechanics, atoms—and therefore all matter—couldn't exist in their stable forms. According to classical physics, electrons orbiting atomic nuclei should rapidly lose energy by radiating electromagnetic waves and spiral into the nucleus, causing all atoms to collapse in a fraction of a second. The quantum mechanical principle that electrons occupy discrete energy levels, and can only transition between these levels by absorbing or emitting specific amounts of energy (quanta), prevents this catastrophic collapse. This quantization of energy—which gives quantum mechanics its name—explains the stability of atoms, the discrete colors emitted by different elements when heated, and forms the basis for understanding all of chemistry.
10. Quantum Coherence Can Exist in Warm, Wet Biological Systems
For decades, scientists believed that quantum effects were too delicate to play any role in biology, requiring extremely cold temperatures and isolated conditions to persist. However, recent research has revealed that quantum coherence—the preservation of quantum states—can occur in living organisms at normal temperatures. Evidence suggests that quantum effects play a role in photosynthesis, where plants apparently use quantum superposition to sample all possible energy pathways simultaneously, allowing them to transfer energy with nearly 100% efficiency. Quantum effects may also be involved in bird navigation, enzyme catalysis, and possibly even consciousness. This emerging field of quantum biology is revealing that life itself may harness quantum mechanics in ways we're only beginning to understand.
Conclusion
These ten facts merely scratch the surface of quantum physics, a field that continues to challenge our understanding of reality while simultaneously enabling revolutionary technologies. From quantum computers that promise to solve problems impossible for classical computers, to quantum cryptography that offers theoretically unbreakable encryption, the practical applications of quantum mechanics are rapidly expanding. Yet perhaps the most profound aspect of quantum physics is how it forces us to reconsider fundamental questions about the nature of reality, observation, and the limits of human knowledge. As research continues and new discoveries emerge, quantum physics remains one of the most exciting and philosophically rich areas of scientific exploration, reminding us that the universe is far stranger and more wonderful than our everyday experience suggests.